


Etudes précliniques pour les maladies inflammatoires de la peau
Les maladies ou symptômes dermatologiques, et notamment l’inflammation cutanée, affectent la vie des patients sur le plan non seulement médical mais aussi social. Les modèles d’inflammation cutanée reposant sur des rongeurs permettent de déchiffrer les mécanismes qui déclenchent et entretiennent la maladie chez les patients.
Oncodesign Services propose plusieurs modèles précliniques standards propres aux pathologies cutanées, et offre le développement de nouveaux modèles d’inflammation cutanée récemment décrits dans la littérature.
Critères d’évaluation pour les maladies inflammatoires
Standards In vivo :
- Score clinique
- Poids corporel
- Grattage en temps réel (prurit)
Standards Ex vivo :
- Score de fibrose
- Examen histopathologique
- Surveillance de biomarqueurs/médicaments
- Expression génique au niveau de la peau, par PCR quantitative/génétique
- Perte d’eau transépidermique
Découvrez les solutions d’Oncodesign Services pour les maladies inflammatoires cutanées
-
Analyses ex vivo
Des explants primaires de peau humaine provenant de donneurs sains sont placés dans une cellule de Franz et traités soit dans le compartiment supérieur pour simuler une application topique, soit dans le compartiment inférieur pour simuler une exposition systémique.
Ils sont généralement utilisés pour analyser les aspects suivants :
- Engagement des cibles
- Fonctionnalité de la barrière cutanée
- Expression de biomarqueurs ou réponse inflammatoire, généralement par PCR.

-
Capacités in vitro
- Kératinocytes primaires humains
- Épreuves : Sécrétion d’IL-8
- Épiderme humain reconstitué (3D)
- Épreuves : Stimulation de voies inflammatoires
- Co-culture avec surnageant bactérien │
- Différenciation │ Prolifération │ Synthèse de triglycérides
- Sébocytes humains
- Épreuves : Synthèse de triglycérides
- Kératinocytes primaires humains
-
Modèles in vivo
Des explants primaires de peau humaine provenant de donneurs sains sont placés dans une cellule de Franz et traités soit dans le compartiment supérieur pour simuler une application topique, soit dans le compartiment inférieur pour simuler une exposition systémique.
Ils sont généralement utilisés pour analyser les aspects suivants :
- Engagement des cibles
- Fonctionnalité de la barrière cutanée
- Expression de biomarqueurs ou réponse inflammatoire, généralement par PCR.
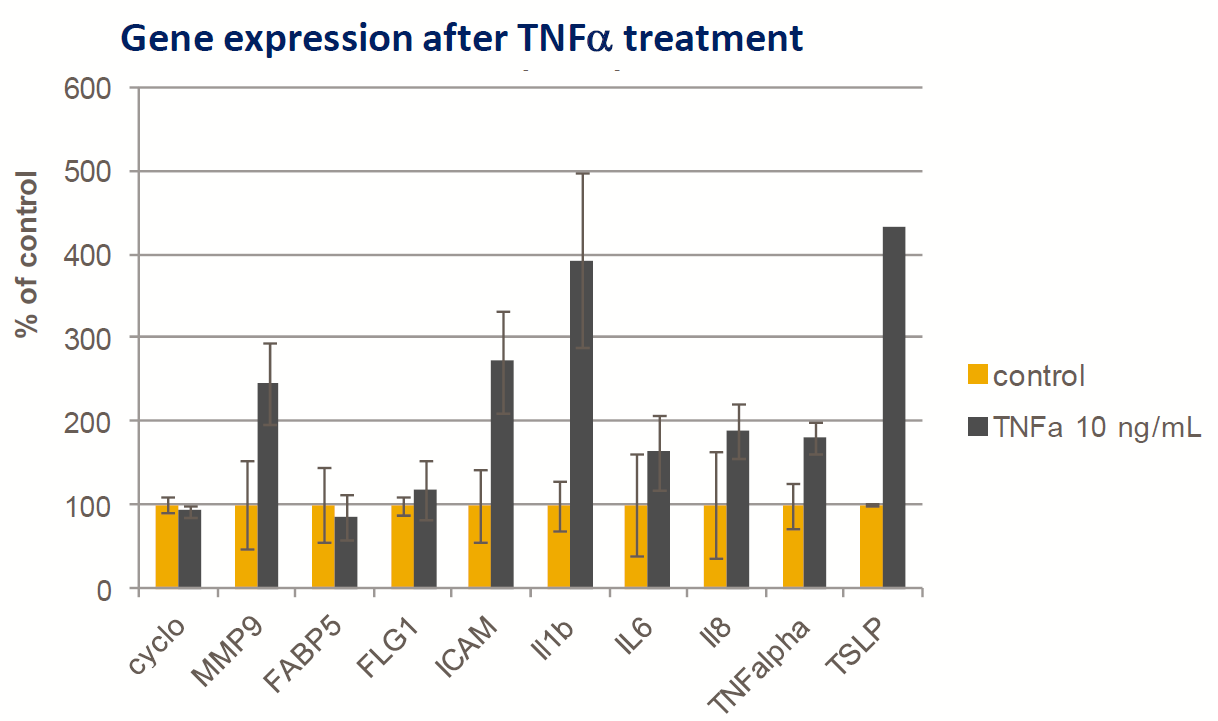
Exemple : biopsie de peau humaine ex vivo
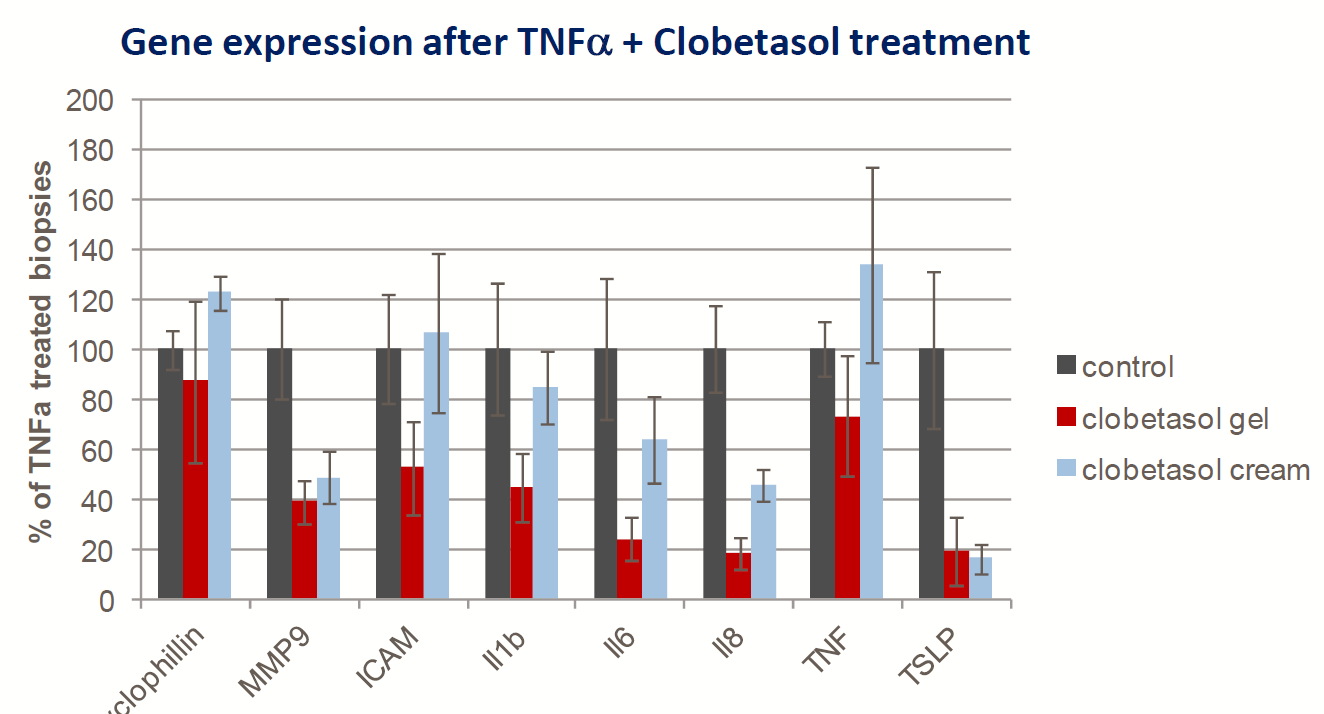
- Biopsie cutanée humaine fraîche
- TNF-α (10 ng/ml) ajouté au compartiment inférieur de la cellule de Franz
- Induction d’une légère réponse génétique inflammatoire (PCR quantitative, technique TaqMan) après 24 h
- Un traitement topique par clobétasol (corticoïde) est capable de réguler négativement l’expression génique induite par le TNF-α (gel > crème)
Etude de cas sur l’inflammation cutanée
-
#1 : Prurit aigu
La voie neuronale impliquée dans la production d’un signal de démangeaison est différente de la voie de transmission de la douleur.
Nos services précliniques pour les modèles de prurit aigu sont basés sur l’inoculation intradermique d’un agent pruritogène :
- Chloroquine
- Substance P
- (de l’imiquimod en application topique peut également être utilisé pour induire le prurit)
La réponse se manifeste généralement dans un délai très bref et est de nature transitoire (< 60 min)
Data acquisition platform used for recording os cratching events (numbers and duration)
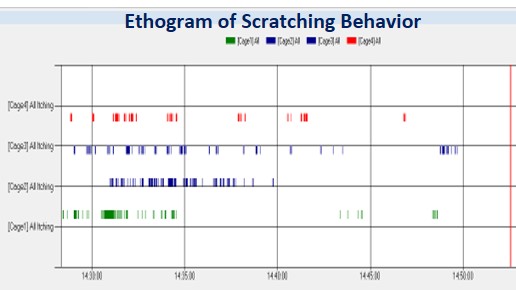

Evaluation of test compounds on itch relief :
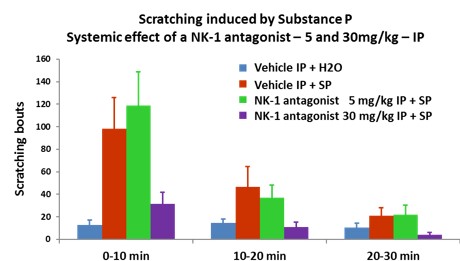
- Test compounds are applied topically 3 hours before itch induction.
- The evaluation of NK-1 antagonist is in the substance P-introduced itch model
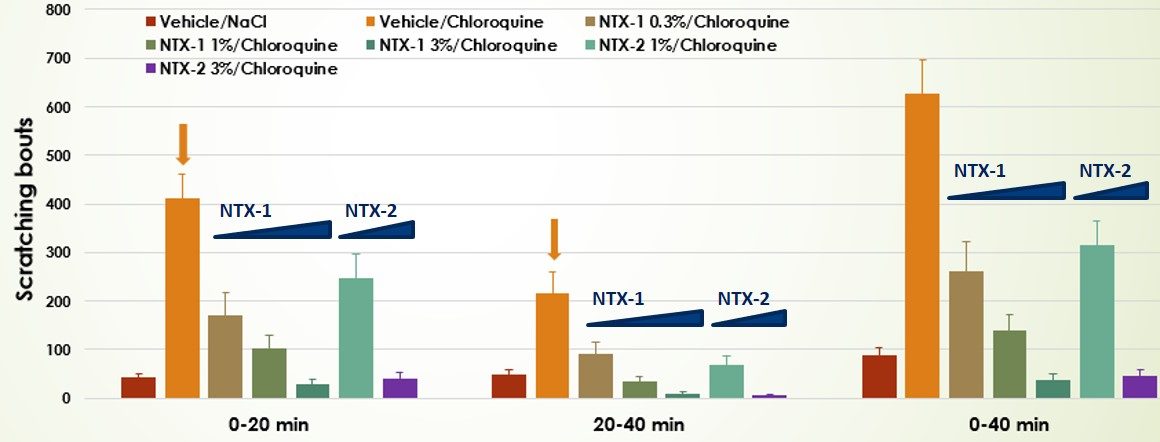
- Dose-response valuation of two test compounds in the Chloroquine-induced itch model
-
#2 : Modèle murin de dermatite atopique induite par des acariens domestiques
L’application topique d’acariens domestiques à des souris BALB/c induit un épaississement de l’épiderme et du derme et le recrutement de cellules inflammatoires (éosinophiles, mastocytes et lymphocytes CD4+).
Ce modèle est utile pour tester l’efficacité de molécules ciblant l’inflammation cutanée générale. Les acariens domestiques sont appliqués directement sur la peau puis la zone est bandée. Le bandage est régulièrement remplacé pendant plusieurs semaines.
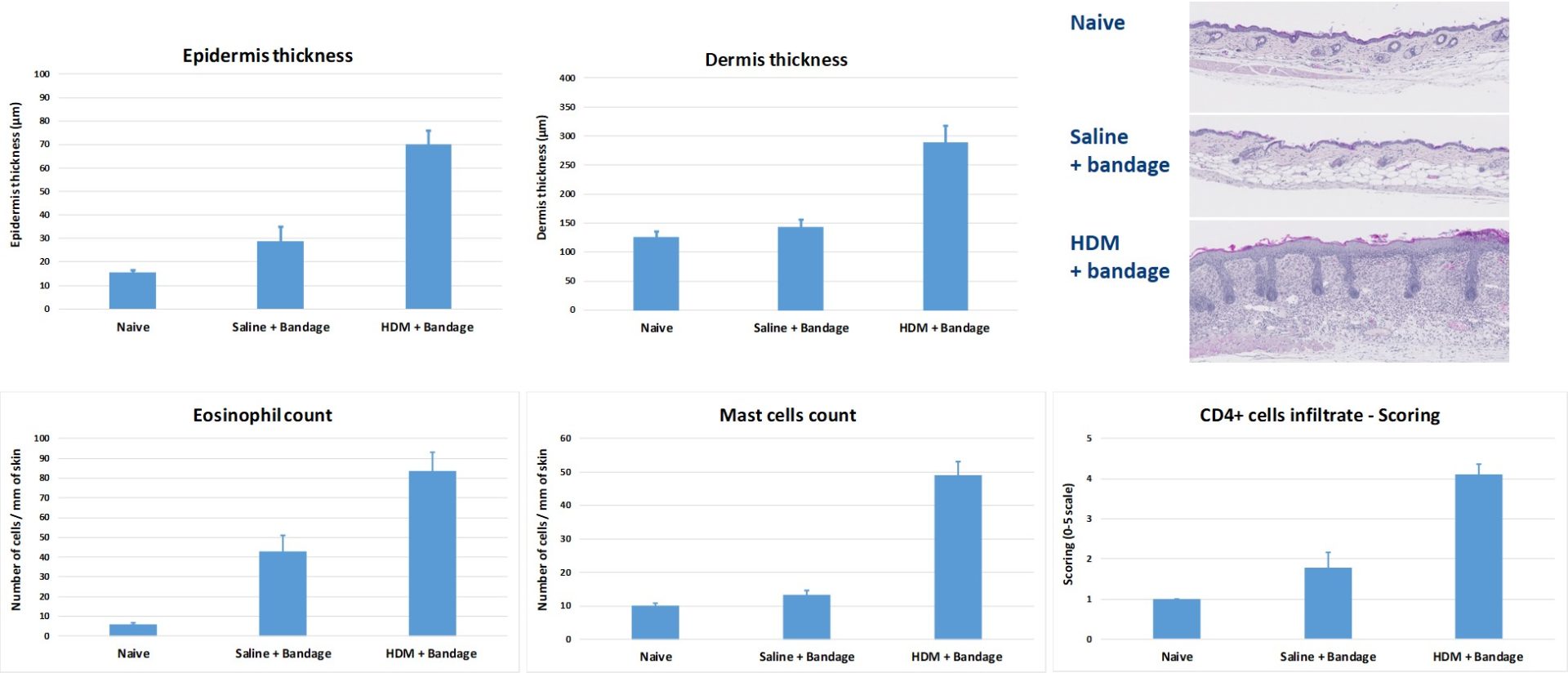
-
#3: Dermatite atopique induite par le calcipotriol chez la souris
L’application topique de calcipotriol à des souris BALB/c (deux fois/jour de J0 à J9) induit des modifications de la morphologie cutanée et une inflammation analogue aux perturbations immunitaires observées dans les lésions aiguës de la dermatite atopique chez des patients humains.
Des médicaments topiques ont montré des effets positifs sur le score d’évaluation clinique de la peau en réduisant l’hyperplasie de l’épiderme et du derme.
- Bétaméthasone (anti-inflammatoire stéroïdien)
- Ruxolinitib (inhibiteur de JAK1/JAK2)
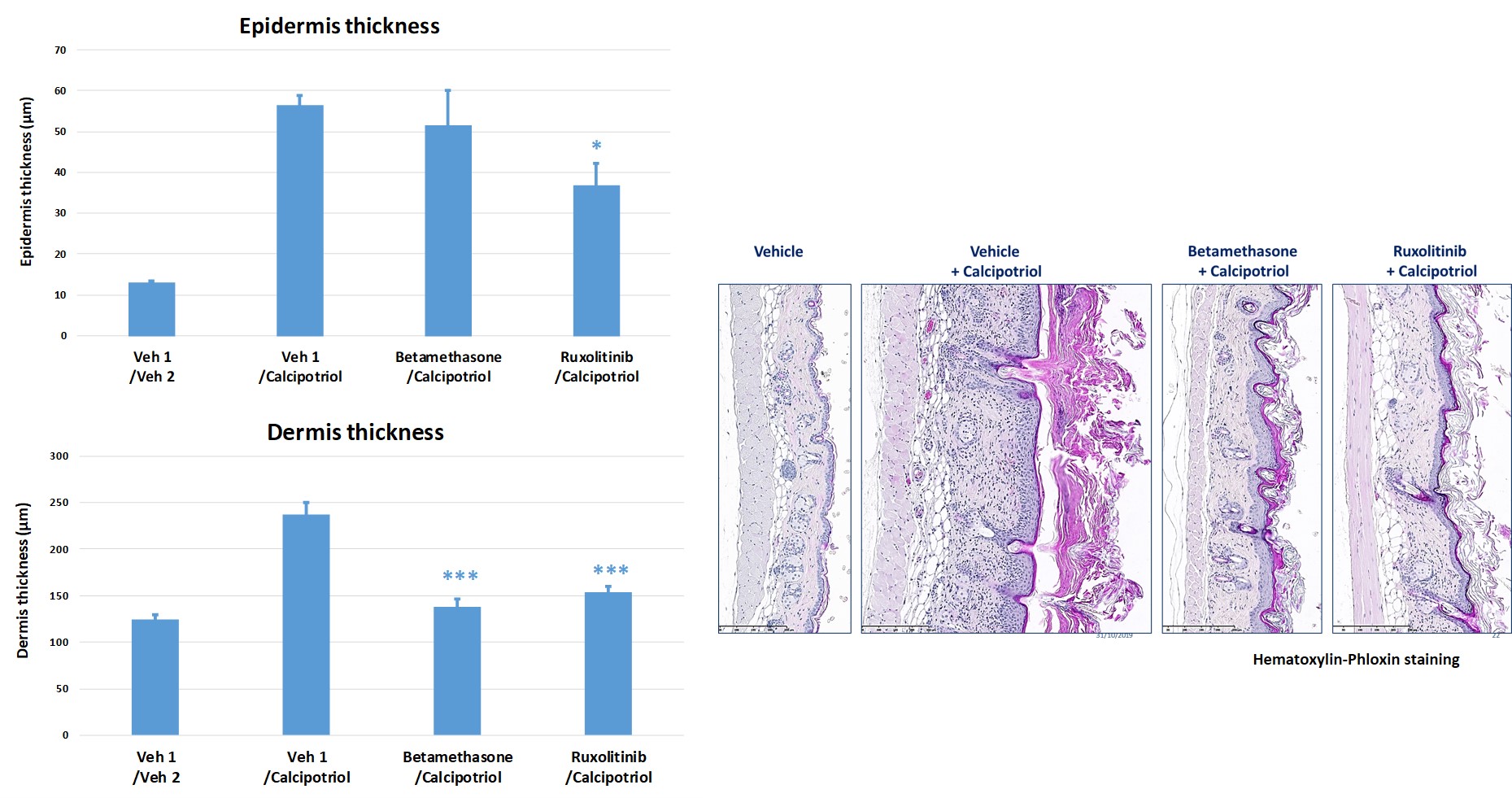
-
#4 : Les médicaments ciblant les lymphocytes Th17 réduisent l’inflammation liée au psoriasis
Le psoriasis peut entraîner un dérèglement de la voie Th17 :
- La différenciation des lymphocytes TH1 à TH17 repose fortement sur le facteur de transcription ROR-γt
- La phosphodiestérase-4 (PDE4) est essentielle pour la libération des interleukines IL-12, IL-23 et du TNF-α
- Des molécules d’essai agissant sur le ROR-γt (antagoniste) et la PDE4 (inhibiteur) ont démontré leur efficacité dans le psoriasis
Les modèles de psoriasis répondent systématiquement aux médicaments oraux et topiques qui montrent une efficacité clinique chez les patients psoriasiques :
- Médicaments oraux: Aprémilast (inhibiteur de la PDE4), fumarate de monométhyle (inhibiteur du NFE2L2), tofacitinib (inhibiteur de JAK)
- Médicaments topiques: Clobétasol (corticoïde), antagoniste du ROR-γt, AN2728 (inhibiteur de la PDE4)
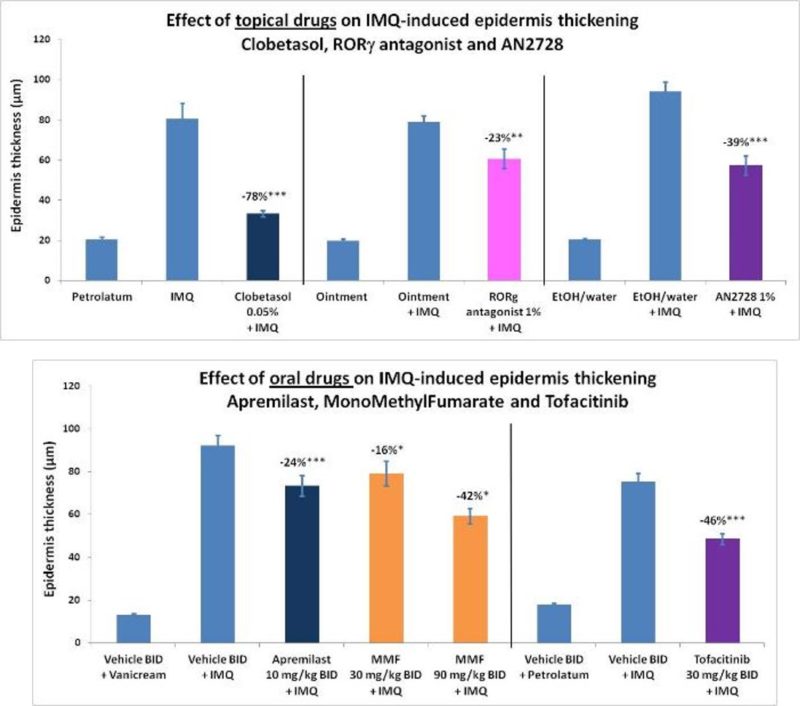
-
Références
(1) Psoriasis model in mice, induced by Imiquimod
- Review:
Gangwar RS, Gudjonsson JE, Ward NL. Mouse Models of Psoriasis: A Comprehensive Review. J Invest Dermatol. 2022 Mar;142(3 Pt B):884-897. doi: 10.1016/j.jid.2021.06.019. Epub 2021 Dec 23. PMID: 34953514. : https://www.jidonline.org/article/S0022-202X(21)01442-1/fulltext
Nițescu DA, Mușetescu A, Nițescu M, Costescu M, Coman OA. Experimental research in topical psoriasis therapy (Review). Exp Ther Med. 2021 Sep;22(3):971. doi: 10.3892/etm.2021.10403. Epub 2021 Jul 8. PMID: 34335913; PMCID: PMC8290406.: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8290406/
- Methods:
Singh TP, Zhang HH, Hwang ST, Farber JM. IL-23- and Imiquimod-Induced Models of Experimental Psoriasis in Mice. Curr Protoc Immunol. 2019 Jun;125(1):e71. doi: 10.1002/cpim.71. Epub 2019 Jan 7. PMID: 30615272. https://currentprotocols.onlinelibrary.wiley.com/doi/10.1002/cpim.71
- FACS in this model (link to FACS resources):
Lou F, Sun Y, Wang H. Protocol for Flow Cytometric Detection of Immune Cell Infiltration in the Epidermis and Dermis of a Psoriasis Mouse Model. STAR Protoc. 2020 Sep 17;1(3):100115. doi: 10.1016/j.xpro.2020.100115. PMID: 33377011; PMCID: PMC7757015. https://www.sciencedirect.com/science/article/pii/S2666166720301027?via%3Dihub
Goldstein JD, Bassoy EY, Caruso A, Palomo J, Rodriguez E, Lemeille S, Gabay C. IL-36 signaling in keratinocytes controls early IL-23 production in psoriasis-like dermatitis. Life Sci Alliance. 2020 Apr 28;3(6):e202000688. doi: 10.26508/lsa.202000688. PMID: 32345660; PMCID: PMC7190273. https://www.life-science-alliance.org/content/3/6/e202000688
- Role of immune system:
Hou Y, Zhu L, Tian H, Sun HX, Wang R, Zhang L, Zhao Y. IL-23-induced macrophage polarization and its pathological roles in mice with imiquimod-induced psoriasis. Protein Cell. 2018 Dec;9(12):1027-1038. doi: 10.1007/s13238-018-0505-z. Epub 2018 Mar 5. PMID: 29508278; PMCID: PMC6251802. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6251802/
van der Fits L, Mourits S, Voerman JS, Kant M, Boon L, Laman JD, Cornelissen F, Mus AM, Florencia E, Prens EP, Lubberts E. Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J Immunol. 2009 May 1;182(9):5836-45. doi: 10.4049/jimmunol.0802999. PMID: 19380832. https://journals.aai.org/jimmunol/article/182/9/5836/104045/Imiquimod-Induced-Psoriasis-Like-Skin-Inflammation
Moos S, Mohebiany AN, Waisman A, Kurschus FC. Imiquimod-Induced Psoriasis in Mice Depends on the IL-17 Signaling of Keratinocytes. J Invest Dermatol. 2019 May;139(5):1110-1117. doi: 10.1016/j.jid.2019.01.006. Epub 2019 Jan 23. PMID: 30684554. https://www.jidonline.org/article/S0022-202X(19)30022-3/fulltext
- Testing of compounds:
Zhang M, Li N, Cai R, Gu J, Xie F, Wei H, Lu C, Wu D. Rosmarinic acid protects mice from imiquimod induced psoriasis-like skin lesions by inhibiting the IL-23/Th17 axis via regulating Jak2/Stat3 signaling pathway. Phytother Res. 2021 Aug;35(8):4526-4537. doi: 10.1002/ptr.7155. Epub 2021 May 18. PMID: 34008239. https://onlinelibrary.wiley.com/doi/10.1002/ptr.7155
Gao J, Chen F, Fang H, Mi J, Qi Q, Yang M. Daphnetin inhibits proliferation and inflammatory response in human HaCaT keratinocytes and ameliorates imiquimod-induced psoriasis-like skin lesion in mice. Biol Res. 2020 Oct 20;53(1):48. doi: 10.1186/s40659-020-00316-0. PMID: 33081840; PMCID: PMC7576854. https://biolres.biomedcentral.com/articles/10.1186/s40659-020-00316-0
Zhang B, Lai RC, Sim WK, Choo ABH, Lane EB, Lim SK. Topical Application of Mesenchymal Stem Cell Exosomes Alleviates the Imiquimod Induced Psoriasis-Like Inflammation. Int J Mol Sci. 2021 Jan 13;22(2):720. doi: 10.3390/ijms22020720. PMID: 33450859; PMCID: PMC7828312. https://www.mdpi.com/1422-0067/22/2/720
Li X, Xi B, Miao Y, Ma X, Zhang J, Gao J, Wei W, Zhou H, Yang C. Nintedanib ameliorates imiquimod-induced psoriasis in mice by inhibiting NF-κB and VEGFR2 signaling. Int Immunopharmacol. 2021 Nov;100:108129. doi: 10.1016/j.intimp.2021.108129. Epub 2021 Sep 20. PMID: 34547680. https://www.sciencedirect.com/science/article/pii/S1567576921007657?via%3Dihub
Kim N, Lee S, Kang J, Choi YA, Jang YH, Jeong GS, Kim SH. Cudraxanthone D Ameliorates Psoriasis-like Skin Inflammation in an Imiquimod-Induced Mouse Model via Inhibiting the Inflammatory Signaling Pathways. Molecules. 2021 Oct 8;26(19):6086. doi: 10.3390/molecules26196086. PMID: 34641629; PMCID: PMC8512696. https://www.mdpi.com/1420-3049/26/19/6086
(2) Psoriasis model in rats, induced by Imiquimod
- Review:
Nițescu DA, Mușetescu A, Nițescu M, Costescu M, Coman OA. Experimental research in topical psoriasis therapy (Review). Exp Ther Med. 2021 Sep;22(3):971. doi: 10.3892/etm.2021.10403. Epub 2021 Jul 8. PMID: 34335913; PMCID: PMC8290406. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8290406/
- Model characterization:
Smajlović A, Haverić A, Alić A, Hadžić M, Smajlović A, Mujezinović I, Lojo-Kadrić N, Ramić J, Elez-Burnjaković N, Haverić S, Pojskić L. Molecular and histopathological profiling of imiquimod induced dermatosis in Swiss Wistar rats: contribution to the rat model for novel anti-psoriasis treatments. Mol Biol Rep. 2021 May;48(5):4295-4303. doi: 10.1007/s11033-021-06445-3. Epub 2021 Jun 7. PMID: 34097205. https://link.springer.com/article/10.1007/s11033-021-06445-3
(3) Skin scleroderma/fibrosis model in mice, induced by Bleomycin
- Review:
Yamamoto T. The bleomycin-induced scleroderma model: what have we learned for scleroderma pathogenesis? Arch Dermatol Res. 2006 Feb;297(8):333-44. doi: 10.1007/s00403-005-0635-z. Epub 2006 Jan 10. PMID: 16402183. https://link.springer.com/article/10.1007/s00403-005-0635-z
Rius Rigau A, Luber M, Distler JHW. Mouse Models of Skin Fibrosis. Methods Mol Biol. 2021;2299:371-383. doi: 10.1007/978-1-0716-1382-5_25. PMID: 34028755. https://link.springer.com/protocol/10.1007/978-1-0716-1382-5_25
- Methods:
Błyszczuk P, Kozlova A, Guo Z, Kania G, Distler O. Experimental Mouse Model of Bleomycin-Induced Skin Fibrosis. Curr Protoc Immunol. 2019 Sep;126(1):e88. doi: 10.1002/cpim.88. PMID: 31483105. https://currentprotocols.onlinelibrary.wiley.com/doi/10.1002/cpim.88
- Involvement of the immune system:
Park MJ, Moon SJ, Lee EJ, Jung KA, Kim EK, Kim DS, Lee JH, Kwok SK, Min JK, Park SH, Cho ML. IL-1-IL-17 Signaling Axis Contributes to Fibrosis and Inflammation in Two Different Murine Models of Systemic Sclerosis. Front Immunol. 2018 Jul 10;9:1611. doi: 10.3389/fimmu.2018.01611. PMID: 30042768; PMCID: PMC6048384. https://www.frontiersin.org/articles/10.3389/fimmu.2018.01611/full
Liu S, Herault Y, Pavlovic G, Leask A. Skin progenitor cells contribute to bleomycin-induced skin fibrosis. Arthritis Rheumatol. 2014 Mar;66(3):707-13. doi: 10.1002/art.38276. PMID: 24574231. https://onlinelibrary.wiley.com/doi/epdf/10.1002/art.38276
Shou Y, Yang L, Yang Y, Xu J. Inhibition of keratinocyte ferroptosis suppresses psoriatic inflammation. Cell Death Dis. 2021 Oct 27;12(11):1009. doi: 10.1038/s41419-021-04284-5. PMID: 34707088; PMCID: PMC8551323. https://www.nature.com/articles/s41419-021-04284-5
- Testing of treatments:
Moon J, Lee SY, Choi JW, Lee AR, Yoo JH, Moon SJ, Park SH, Cho ML. Metformin ameliorates scleroderma via inhibiting Th17 cells and reducing mTOR-STAT3 signaling in skin fibroblasts. J Transl Med. 2021 May 4;19(1):192. doi: 10.1186/s12967-021-02860-z. Erratum in: J Transl Med. 2021 Jun 21;19(1):266. PMID: 33947424; PMCID: PMC8097822. https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-021-02860-z
Li R, Yin H, Wang J, He D, Yan Q, Lu L. Dihydroartemisinin alleviates skin fibrosis and endothelial dysfunction in bleomycin-induced skin fibrosis models. Clin Rheumatol. 2021 Oct;40(10):4269-4277. doi: 10.1007/s10067-021-05765-w. Epub 2021 May 19. PMID: 34013490. https://link.springer.com/article/10.1007/s10067-021-05765-w
(4) Skin scleroderma/fibrosis model in mice, induced by Topoisomerase-I peptide-loaded dendritic cells
- Review:
Rius Rigau A, Luber M, Distler JHW. Mouse Models of Skin Fibrosis. Methods Mol Biol. 2021;2299:371-383. doi: 10.1007/978-1-0716-1382-5_25. PMID: 34028755. https://link.springer.com/protocol/10.1007/978-1-0716-1382-5_25
- Model characterization:
Mehta H, Goulet PO, Nguyen V, Pérez G, Koenig M, Senécal JL, Sarfati M. Topoisomerase I peptide-loaded dendritic cells induce autoantibody response as well as skin and lung fibrosis. Autoimmunity. 2016 Dec;49(8):503-513. doi: 10.1080/08916934.2016.1230848. Epub 2016 Nov 3. PMID: 27808577. https://www.tandfonline.com/doi/abs/10.1080/08916934.2016.1230848?journalCode=iaut20
- Role of microbiome
Mehta H, Goulet PO, Mashiko S, Desjardins J, Pérez G, Koenig M, Senécal JL, Constante M, Santos MM, Sarfati M. Early-Life Antibiotic Exposure Causes Intestinal Dysbiosis and Exacerbates Skin and Lung Pathology in Experimental Systemic Sclerosis. J Invest Dermatol. 2017 Nov;137(11):2316-2325. doi: 10.1016/j.jid.2017.06.019. Epub 2017 Jul 27. PMID: 28757138. https://www.jidonline.org/article/S0022-202X(17)31854-7/fulltext
Ho KJ, Varga J. Early-Life Gut Dysbiosis: A Driver of Later-Life Fibrosis? J Invest Dermatol. 2017 Nov;137(11):2253-2255. doi: 10.1016/j.jid.2017.08.017. PMID: 29055411. https://www.jidonline.org/article/S0022-202X(17)32831-2/fulltext
(5) Skin scleroderma/fibrosis model in mice, induced by Topoisomerase-I and CFA
- Review:
Rius Rigau A, Luber M, Distler JHW. Mouse Models of Skin Fibrosis. Methods Mol Biol. 2021;2299:371-383. doi: 10.1007/978-1-0716-1382-5_25. PMID: 34028755. https://link.springer.com/protocol/10.1007/978-1-0716-1382-5_25
Yoshizaki A, Yanaba K, Ogawa A, Asano Y, Kadono T, Sato S. Immunization with DNA topoisomerase I and Freund’s complete adjuvant induces skin and lung fibrosis and autoimmunity via interleukin-6 signaling. Arthritis Rheum. 2011 Nov;63(11):3575-85. doi: 10.1002/art.30539. PMID: 21792823. https://onlinelibrary.wiley.com/doi/epdf/10.1002/art.30539
(6) Atopic dermatitis model in mice, induced by Calcipotriol
- Reviews:
Guerrero-Aspizua S, Carretero M, Conti CJ, Del Río M. The importance of immunity in the development of reliable animal models for psoriasis and atopic dermatitis. Immunol Cell Biol. 2020 Sep;98(8):626-638. doi: 10.1111/imcb.12365. Epub 2020 Jul 15. PMID: 32479655. https://onlinelibrary.wiley.com/doi/10.1111/imcb.12365
Kim D, Kobayashi T, Nagao K. Research Techniques Made Simple: Mouse Models of Atopic Dermatitis. J Invest Dermatol. 2019 May;139(5):984-990.e1. doi: 10.1016/j.jid.2019.02.014. Epub 2019 Apr 19. PMID: 31010529; PMCID: PMC6555635. https://www.jidonline.org/article/S0022-202X(19)30184-8/fulltext
- Methods:
Moosbrugger-Martinz V, Schmuth M, Dubrac S. A Mouse Model for Atopic Dermatitis Using Topical Application of Vitamin D3 or of Its Analog MC903. Methods Mol Biol. 2017;1559:91-106. doi: 10.1007/978-1-4939-6786-5_8. PMID: 28063040. https://link.springer.com/protocol/10.1007/978-1-4939-6786-5_8
- Immune mechanisms:
Wan H, Yang H, Wei M, Chen W. Polyinosinic:polycytidylic acid aggravates calcipotriol-induced atopic dermatitis-like skin lesions in mice by increasing the expression of thymic stromal lymphopoietin. Ann Transl Med. 2022 Feb;10(4):209. doi: 10.21037/atm-22-282. PMID: 35280398; PMCID: PMC8908153. https://atm.amegroups.com/article/view/90304/html
Li M, Hener P, Zhang Z, Kato S, Metzger D, Chambon P. Topical vitamin D3 and low-calcemic analogs induce thymic stromal lymphopoietin in mouse keratinocytes and trigger an atopic dermatitis. Proc Natl Acad Sci U S A. 2006 Aug 1;103(31):11736-41. doi: 10.1073/pnas.0604575103. Epub 2006 Jul 31. PMID: 16880407; PMCID: PMC1544239. https://www.pnas.org/doi/10.1073/pnas.0604575103?url_ver=Z39.88-2003&rfr_id=ori%3Arid%3Acrossref.org&rfr_dat=cr_pub++0pubmed
Li M, Hener P, Zhang Z, Ganti KP, Metzger D, Chambon P. Induction of thymic stromal lymphopoietin expression in keratinocytes is necessary for generating an atopic dermatitis upon application of the active vitamin D3 analogue MC903 on mouse skin. J Invest Dermatol. 2009 Feb;129(2):498-502. doi: 10.1038/jid.2008.232. Epub 2008 Jul 24. PMID: 18650845. https://www.jidonline.org/article/S0022-202X(15)34183-X/fulltext
- Compound testing :
Seshimo H, Egusa C, Maeda T, Numata T, Okubo Y, Harada K, Ito T. Topical application of imatinib mesylate suppresses vitamin D3 analog-induced dermatitis in Balb/c mice. Exp Dermatol. 2022 Dec 1. doi: 10.1111/exd.14720. Epub ahead of print. PMID: 36457228. https://onlinelibrary.wiley.com/doi/10.1111/exd.14720
(7) Atopic dermatitis model in mice, induced by DNFB
- Reviews:
Guerrero-Aspizua S, Carretero M, Conti CJ, Del Río M. The importance of immunity in the development of reliable animal models for psoriasis and atopic dermatitis. Immunol Cell Biol. 2020 Sep;98(8):626-638. doi: 10.1111/imcb.12365. Epub 2020 Jul 15. PMID: 32479655. https://onlinelibrary.wiley.com/doi/10.1111/imcb.12365
Kim D, Kobayashi T, Nagao K. Research Techniques Made Simple: Mouse Models of Atopic Dermatitis. J Invest Dermatol. 2019 May;139(5):984-990.e1. doi: 10.1016/j.jid.2019.02.014. Epub 2019 Apr 19. PMID: 31010529; PMCID: PMC6555635. https://www.jidonline.org/article/S0022-202X(19)30184-8/fulltext
- Testing of compounds:
Tang L, Gao J, Cao X, Chen L, Wang H, Ding H. TRPV1 mediates itch-associated scratching and skin barrier dysfunction in DNFB-induced atopic dermatitis mice. Exp Dermatol. 2022 Mar;31(3):398-405. doi: 10.1111/exd.14464. Epub 2021 Oct 11. PMID: 34608683. https://onlinelibrary.wiley.com/doi/10.1111/exd.14464
Liu Q, Li M, Wang N, He C, Jiang X, Li J. Calcium-Based Antimicrobial Peptide Compounds Attenuate DNFB-Induced Atopic Dermatitis-Like Skin Lesions via Th-Cells in BALB/c Mice. Int J Mol Sci. 2022 Sep 26;23(19):11371. doi: 10.3390/ijms231911371. PMID: 36232673; PMCID: PMC9569644. https://www.mdpi.com/1422-0067/23/19/11371
Gao JF, Tang L, Luo F, Zhang YY, Chen L, Ding H, Meng ZD. Nicotinamide mononucleotide ameliorates DNFB-induced atopic dermatitis-like symptoms in mice by blocking activation of ROS-mediated JAK2/STAT5 signaling pathway. Int Immunopharmacol. 2022 Aug;109:108812. doi: 10.1016/j.intimp.2022.108812. Epub 2022 May 6. PMID: 35533554. https://www.sciencedirect.com/science/article/pii/S156757692200296X?via%3Dihub
(8) Atopic dermatitis model in mice, induced by HDM
- Reviews:
Guerrero-Aspizua S, Carretero M, Conti CJ, Del Río M. The importance of immunity in the development of reliable animal models for psoriasis and atopic dermatitis. Immunol Cell Biol. 2020 Sep;98(8):626-638. doi: 10.1111/imcb.12365. Epub 2020 Jul 15. PMID: 32479655 https://onlinelibrary.wiley.com/doi/10.1111/imcb.12365
Kim D, Kobayashi T, Nagao K. Research Techniques Made Simple: Mouse Models of Atopic Dermatitis. J Invest Dermatol. 2019 May;139(5):984-990.e1. doi: 10.1016/j.jid.2019.02.014. Epub 2019 Apr 19. PMID: 31010529; PMCID: PMC6555635. https://www.jidonline.org/article/S0022-202X(19)30184-8/fulltext
- Mechanism:
Lee YS, Choi JH, Lee JH, Lee HW, Lee W, Kim WT, Kim TY. Extracellular superoxide dismutase ameliorates house dust mite-induced atopic dermatitis-like skin inflammation and inhibits mast cell activation in mice. Exp Dermatol. 2016 Aug;25(8):630-5. doi: 10.1111/exd.13028. Epub 2016 Jun 30. PMID: 27061078. https://onlinelibrary.wiley.com/doi/10.1111/exd.13028
(9) Pruritogen itch model in mice, induced by Chloroquine, Substance P or Imiquimod
- Scratching measurement and comparison of mouse strains:
Sargent JL, Löhr CV, Diggs HE. Scratching Responses to Epidermal Injury in C57BL/6, DBA/2, BALB/c, and CD1 Mice. Comp Med. 2016;66(3):208-15. PMID: 27298245; PMCID: PMC4907529. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4907529/
- Chloroquine:
Liu Q, Tang Z, Surdenikova L, Kim S, Patel KN, Kim A, Ru F, Guan Y, Weng HJ, Geng Y, Undem BJ, Kollarik M, Chen ZF, Anderson DJ, Dong X. Sensory neuron-specific GPCR Mrgprs are itch receptors mediating chloroquine-induced pruritus. Cell. 2009 Dec 24;139(7):1353-65. doi: 10.1016/j.cell.2009.11.034. Epub 2009 Dec 10. PMID: 20004959; PMCID: PMC2989405. https://www.cell.com/cell/fulltext/S0092-8674(09)01492-5
Shiraishi Y, Koga K, Yamagata R, Hatada I, Shiratori-Hayashi M, Tsuda M. α1A-adrenaline receptors in dorsal horn inhibitory neurons have an inhibitory role in the regulation of chloroquine-induced itch in mice. Mol Brain. 2021 Mar 16;14(1):55. doi: 10.1186/s13041-021-00768-9. PMID: 33726812; PMCID: PMC7962300. https://molecularbrain.biomedcentral.com/articles/10.1186/s13041-021-00768-9
- Chloroquine itch and microbiota:
Zhang Q, Li T, Niu J, Xiao J, Zhang M, Zhang R, Chen D, Shi Y, Zhang X, Hu X, Yu B, Feng J, Fang Q. Inhibitory effects of antibiotic-induced gut microbiota depletion on acute itch behavior in mice. Brain Res Bull. 2022 Nov;190:50-61. doi: 10.1016/j.brainresbull.2022.09.014. Epub 2022 Sep 17. PMID: 36126873. https://www.sciencedirect.com/science/article/abs/pii/S0361923022002544?via%3Dihub
Li R, Sun H, Zheng H, Zong Z, Li S, Meng T, Li J, Liu Y, Wang C, Li J. Intradermal Injection of Oxytocin Aggravates Chloroquine-Induced Itch Responses via Activating the Vasopressin-1a Receptor/Nitric Oxide Pathway in Mice. Front Pharmacol. 2019 Nov 15;10:1380. doi: 10.3389/fphar.2019.01380. PMID: 31824317; PMCID: PMC6881818. https://www.frontiersin.org/articles/10.3389/fphar.2019.01380/full
- Substance P:
Andoh T, Katsube N, Maruyama M, Kuraishi Y. Involvement of leukotriene B(4) in substance P-induced itch-associated response in mice. J Invest Dermatol. 2001 Dec;117(6):1621-6. doi: 10.1046/j.0022-202x.2001.01585.x. PMID: 11886531. https://www.jidonline.org/article/S0022-202X(15)41505-2/fulltext
Azimi E, Reddy VB, Pereira PJS, Talbot S, Woolf CJ, Lerner EA. Substance P activates Mas-related G protein-coupled receptors to induce itch. J Allergy Clin Immunol. 2017 Aug;140(2):447-453.e3. doi: 10.1016/j.jaci.2016.12.980. Epub 2017 Feb 20. PMID: 28219706; PMCID: PMC5546940. https://www.jacionline.org/article/S0091-6749(17)30230-0/fulltext
Andoh T, Kuraishi Y. Nitric oxide enhances substance P-induced itch-associated responses in mice. Br J Pharmacol. 2003 Jan;138(1):202-8. doi: 10.1038/sj.bjp.0705004. PMID: 12522091; PMCID: PMC1573631. https://bpspubs.onlinelibrary.wiley.com/doi/full/10.1038/sj.bjp.0705004
Akasaka Y, Yoshida T, Tsukahara M, Hatta A, Inoue H. Glycyrrhetinic acid prevents cutaneous scratching behavior in mice elicited by substance P or PAR-2 agonist. Eur J Pharmacol. 2011 Nov 16;670(1):175-9. doi: 10.1016/j.ejphar.2011.08.043. Epub 2011 Sep 10. PMID: 21925497. https://www.sciencedirect.com/science/article/abs/pii/S0014299911009538?via%3Dihub
Andoh T, Kuraishi Y. Inhibitory effects of azelastine on substance P-induced itch-associated response in mice. Eur J Pharmacol. 2002 Feb 2;436(3):235-9. doi: 10.1016/s0014-2999(01)01617-x. PMID: 11858803. https://www.sciencedirect.com/science/article/abs/pii/S001429990101617X?via%3Dihub
- Imiquimod-induced itch:
Sakai K, Sanders KM, Youssef MR, Yanushefski KM, Jensen L, Yosipovitch G, Akiyama T. Mouse model of imiquimod-induced psoriatic itch. Pain. 2016 Nov;157(11):2536-2543. doi: 10.1097/j.pain.0000000000000674. PMID: 27437787; PMCID: PMC5069152. https://journals.lww.com/pain/Abstract/2016/11000/Mouse_model_of_imiquimod_induced_psoriatic_itch.19.aspx
Li L, Liu X, Ge W, Chen C, Huang Y, Jin Z, Zhan M, Duan X, Liu X, Kong Y, Jiang J, Li X, Zeng X, Li F, Xu S, Li M, Chen H. CB2R Deficiency Exacerbates Imiquimod-Induced Psoriasiform Dermatitis and Itch Through the Neuro-Immune Pathway. Front Pharmacol. 2022 Jan 31;13:790712. doi: 10.3389/fphar.2022.790712. PMID: 35173615; PMCID: PMC8841964. https://www.frontiersin.org/articles/10.3389/fphar.2022.790712/full
Xu Z, Qin Z, Zhang J, Wang Y. Microglia-mediated chronic psoriatic itch induced by imiquimod. Mol Pain. 2020 Jan-Dec;16:1744806920934998. doi: 10.1177/1744806920934998. PMID: 32580615; PMCID: PMC7318815. https://journals.sagepub.com/doi/full/10.1177/1744806920934998
(10) Acnea model in rats and mice, induced by Sebaceous gland atrophy
Meingassner JG, Aschauer H, Winiski AP, Dales N, Yowe D, Winther MD, Zhang Z, Stütz A, Billich A. Pharmacological inhibition of stearoyl CoA desaturase in the skin induces atrophy of the sebaceous glands. J Invest Dermatol. 2013 Aug;133(8):2091-4. doi: 10.1038/jid.2013.89. Epub 2013 Feb 27. PMID: 23446987. https://www.jidonline.org/article/S0022-202X(15)36362-4/fulltext
Shang W, Tan AYQ, van Steensel MAM, Lim X. Aberrant Wnt Signaling Induces Comedo-Like Changes in the Murine Upper Hair Follicle. J Invest Dermatol. 2022 Oct;142(10):2603-2612.e6. doi: 10.1016/j.jid.2021.11.034. Epub 2021 Dec 17. PMID: 34929175. https://www.jidonline.org/article/S0022-202X(21)02616-6/fulltext
Case studies for skin inflammation:
- #1 : Acute Itch : Same refs as above
- #2 : House Dust Mite (HDM)-Atopic Dermatitis (AD) mouse model : Same refs as above
- #3: Calcipotriol-induced atopic dermatitis in mice : Same refs as above
- #4: TH17 cell-targeting drugs reduce psoriasis-related inflammation
Zhang M, Li N, Cai R, Gu J, Xie F, Wei H, Lu C, Wu D. Rosmarinic acid protects mice from imiquimod induced psoriasis-like skin lesions by inhibiting the IL-23/Th17 axis via regulating Jak2/Stat3 signaling pathway. Phytother Res. 2021 Aug;35(8):4526-4537. doi: 10.1002/ptr.7155. Epub 2021 May 18. PMID: 34008239. https://onlinelibrary.wiley.com/doi/10.1002/ptr.7155
Shou Y, Yang L, Yang Y, Xu J. Inhibition of keratinocyte ferroptosis suppresses psoriatic inflammation. Cell Death Dis. 2021 Oct 27;12(11):1009. doi: 10.1038/s41419-021-04284-5. PMID: 34707088; PMCID: PMC8551323. https://www.nature.com/articles/s41419-021-04284-5
