Endometriosis Awareness Month places a necessary spotlight on a disease that is often under-recognized and poorly understood. For researchers, it is also a reminder of the complexity that emerges long before a therapy reaches patients. Preclinical endometriosis research sits at the intersection of functional disease, unique biology, and translational uncertainty, making careful model and endpoint selection essential.
What makes preclinical endometriosis research so challenging?
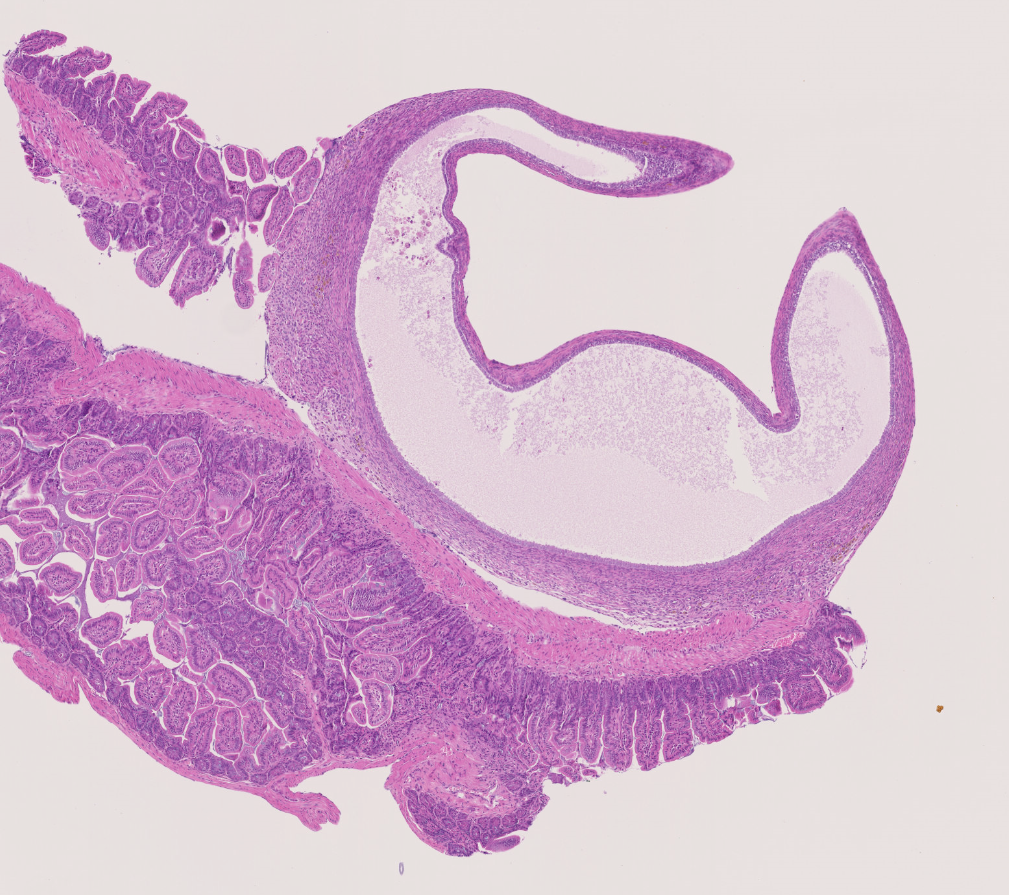
Endometriosis is often described as a complex disease, and that complexity becomes especially apparent in preclinical research. Long before a therapy reaches the clinic, researchers are faced with a core problem: the biology that defines endometriosis in patients does not translate neatly into experimental systems.
At a high level, preclinical endometriosis research is challenging because:
- Key patient outcomes such as infertility are difficult to model
- Menstruation plays a central biological role that rodents do not replicate
- The disease burden is largely functional, particularly pain
- Clinical symptoms do not map directly onto standard research model endpoints
- No single model captures the full disease or answers all translational questions
In patients, endometriosis burden is typically defined less by visible pathology and more by lived experience. Pain dominates, often alongside infertility and long-term impacts on quality of life. These features explain why treatment is needed, but they are difficult to capture using conventional preclinical readouts such as lesion size, histology, or inflammatory markers.
Biology adds another layer of complexity. Endometriosis is closely linked to menstruation, yet mice do not menstruate. This fundamental difference means that even well-established models can only approximate certain aspects of disease initiation and progression, rather than fully recapitulate the human condition.
This is where model choice becomes critical. To better reflect human biology, some preclinical approaches rely on lesions obtained directly from patients and engrafted into immunocompromised models, allowing disease-relevant tissue architecture and cellular composition to be preserved. These strategies can improve clinical relevance, but they also introduce variability and experimental constraints that must be carefully managed.
Humanized systems offer another way to bridge the translational gap. Models humanised for a specific therapeutic target, or for components of the human immune system, can enable more meaningful assessment of target engagement and mechanism of action. However, humanization does not eliminate complexity; these models still require careful interpretation and are most powerful when tightly aligned with a clear biological hypothesis.
Finally, standard preclinical approaches often rely on static, single time-point measurements. These can miss disease dynamics, treatment kinetics, and functional effects that evolve over time, all of which are highly relevant in a chronic condition like endometriosis.
In practice, progress in preclinical endometriosis research depends less on finding a “perfect” model, and more on a holistic approach that carefully aligns biology, model design, and readouts with the clinical question being asked.