


Studying the pharmacokinetics and metabolism (DMPK/ADME) of your new therapeutic entities
The DMPK department at Oncodesign Services offers in vitro, in life and bioanalytical components to provide complete packages for:
- ad hoc in vitro ADME-T prediction studies and drug metabolism studies
- the design of an integrated set of in vitro and in vivo assays and decision trees tailored to the different stages of progression of your discovery program, from target to hit to preclinical candidate selection, and therapeutic dose prediction in humans.
- pharmacokinetic (PK) profile and PK/PD relationship assessment studies in rodents (including pathological models) and other animal species (drug exposure and disease-related)
- biodistribution with radiolabeled test articles using radioisotopes: 3H, 14C, 125I
- De-risking DMPK packages customized according to your data and needs
- Introduction to non-invasive biodistribution of drugs using imaging capabilities (SPECT/TEP) in rodents, NHPs.
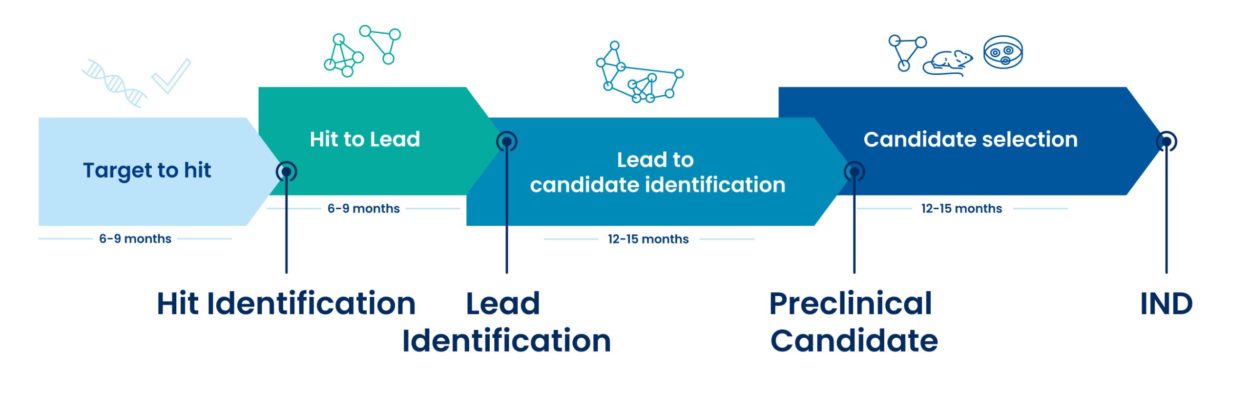
ADME/DMPK Assays are recommended at each stage of drug discovery, to de-risk the compound in cost-effective manner
-
Target to hit
Evaluation of physicochemical and in vitro ADME properties
- Solubility
- LogD
- Microsomal strability
- CYP inhibition
-
Hit to Lead
Optimization of physicochemical and drug-like properties
- Metabolic stability
- CYP inhibition
- Permeability
- In vitro plasma protein binding
- Combined PK-BBB
- Unbound Partition Coefficient (Kpu,u brain)
-
Lead to candidate identification
Optimization of drug-like properties, IVIVC, PK/PD correlation
- Permeability
- In vitro plasma protein binding
- PK studies (rodents & non-rodents)
- Target tissue exposure
- Blood/plasma partitioning
- Mass balance
- PK/PD studies
- Metabolites identification
-
Candidate selection
Dose range finding, safety/tox evaluation, human dose prediction
- Dose range finding (rodents & non-rodents)
- Tissue distribution
- Food effect
- Metabolite profiling
- Interspecies scoring
- Metabolites identification
- CYP induction
Do you have a request for DMPK services ? Contact-us !


