Why Choose Humanized Mouse Models for Oncology & Inflammation Research?
At Oncodesign Services, our advanced humanized mouse models serve as versatile platforms for translational research in oncology and inflammatory disease.
Engineered with components of the human immune system, these humanized mice mimic tumor-immune interactions and inflammatory responses, making them essential for evaluating immunotherapies, anti-inflammatory agents, and combination strategies. Whether your focus is cancer immunotherapy or chronic inflammatory pathways, our customizable humanized mice models deliver high impact preclinical data.
Pioneer in humanization since 2002
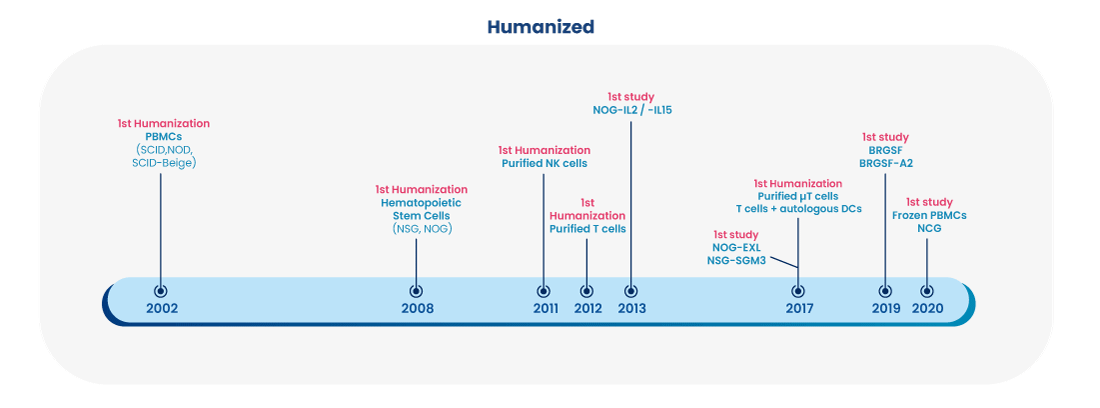
As early as 2002, Oncodesign Services began using humanized model to support the study of antibodies, bispecifics, CAR-Ts and other immuno-modulatory therapies requiring a human immune system. Humanized mice can be either genetically modified straits for the expression of specific human genes, or immunodeficient mice receiving a human cell transplantation (e.g. using peripheral blood mononuclear cells or human CD34+ hematopietic stem cells).
We continue to adopt the latest approaches to humanization, so as to remain the leading CRO expert offering humanized mouse study services for preclinical pharmacology modeling.
Typical readouts for immuno-oncology studies with humanized mice models:
- Tumor size
- Survival
- Cytokine/biomarker expression (by LBA)
- TIL fractions (by flow/FACS)
Building Humanized mouse models
Oncodesign Services is agnostic to approach, selecting the right humanized model for the right study. Humanization of rodents (mice, rats) can be approached at two levels:
- Cellular humanization corresponds to xenograft transplantation of human cells or tissues, to provide a source of human leucocytes (CD34+ hematopoietic stem cells, PBMC, purified leucocyte sub-populations)
- Genetic humanization corresponds to mouse gene swap by their human equivalent, either to provide a relevant target to a human-specific therapy, or endogenous support to the generation, maintenance and function of transplanted human cells
A large variety of mouse recipient models are available (nude mice, scid mice, NOD scid mice, NSG mice, NOG mice, BRGSF mice, NCG mice, immunodeficient rats, etc.) from providers. Depending on the human leucocyte subset of interest, specific models might provide specific advantages – for instance, BRGSF mice permit an improved accumulation of human myeloid cells after HSC transplantation, with the possibility to transiently boost the human dendritic cell compartment.
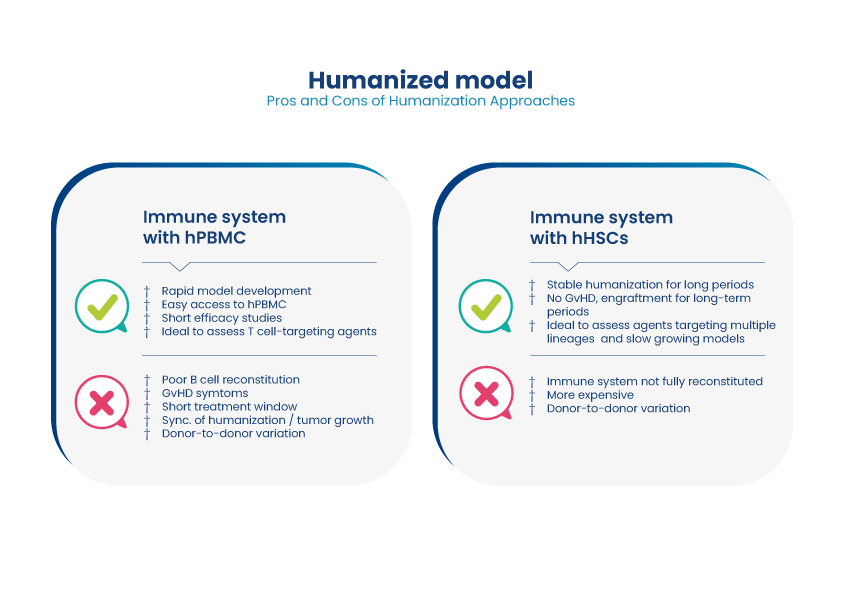
Case studies: Application of Humanized Mouse Models in Oncology and Inflammation
Case study #1 – PBMCs in NOG mice
In this study, Oncodesign Services tested a BiTe antibody fragment in the B-cell lymphoma line Ramos BCL, humanized with PBMCs in NOG mice. The treatment successfully increased survival, with tumor cell depletion demonstrated by flow/FACS analysis of sampled blood.
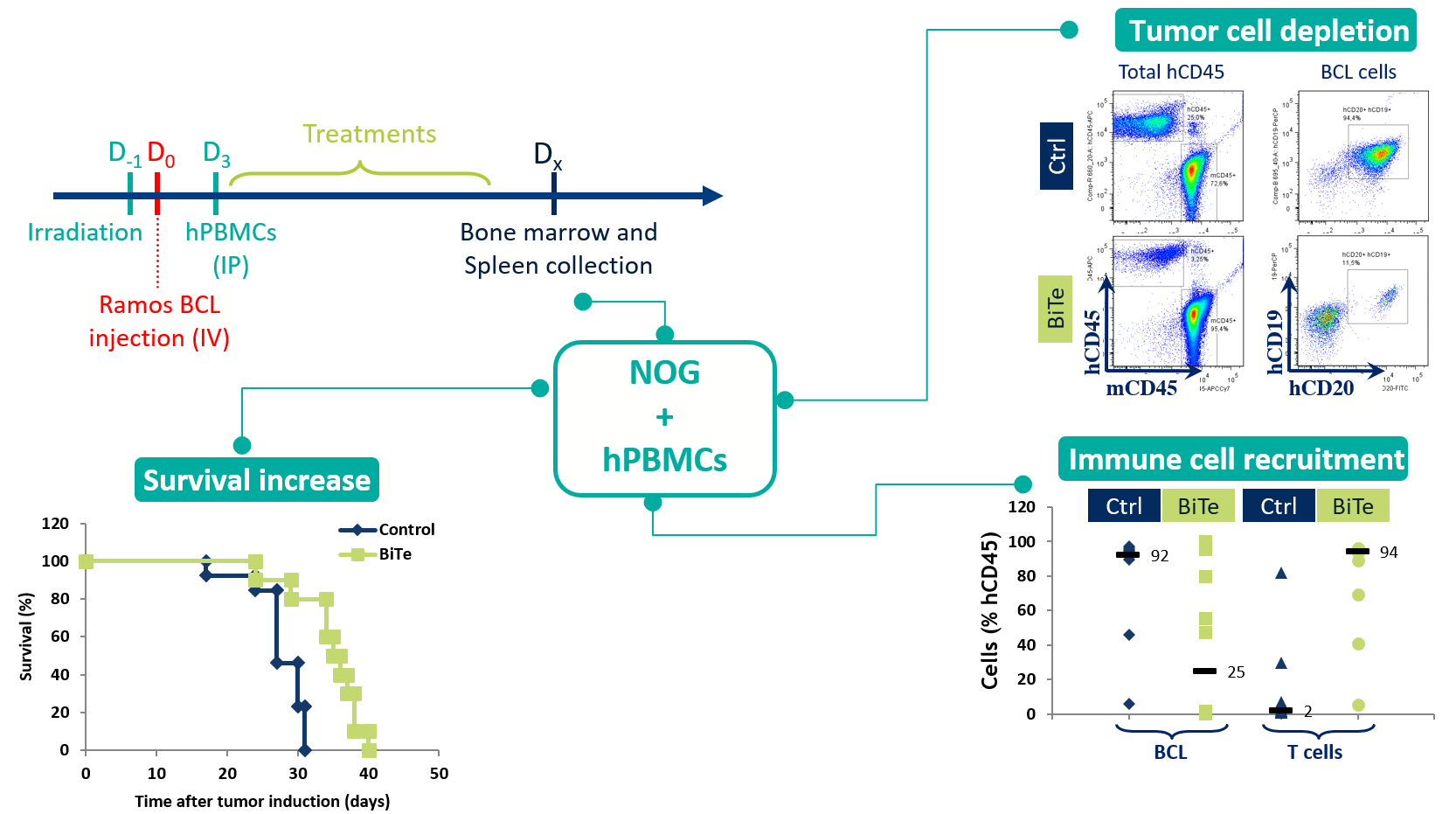
Case study #2 – PBMCs in NOG mice
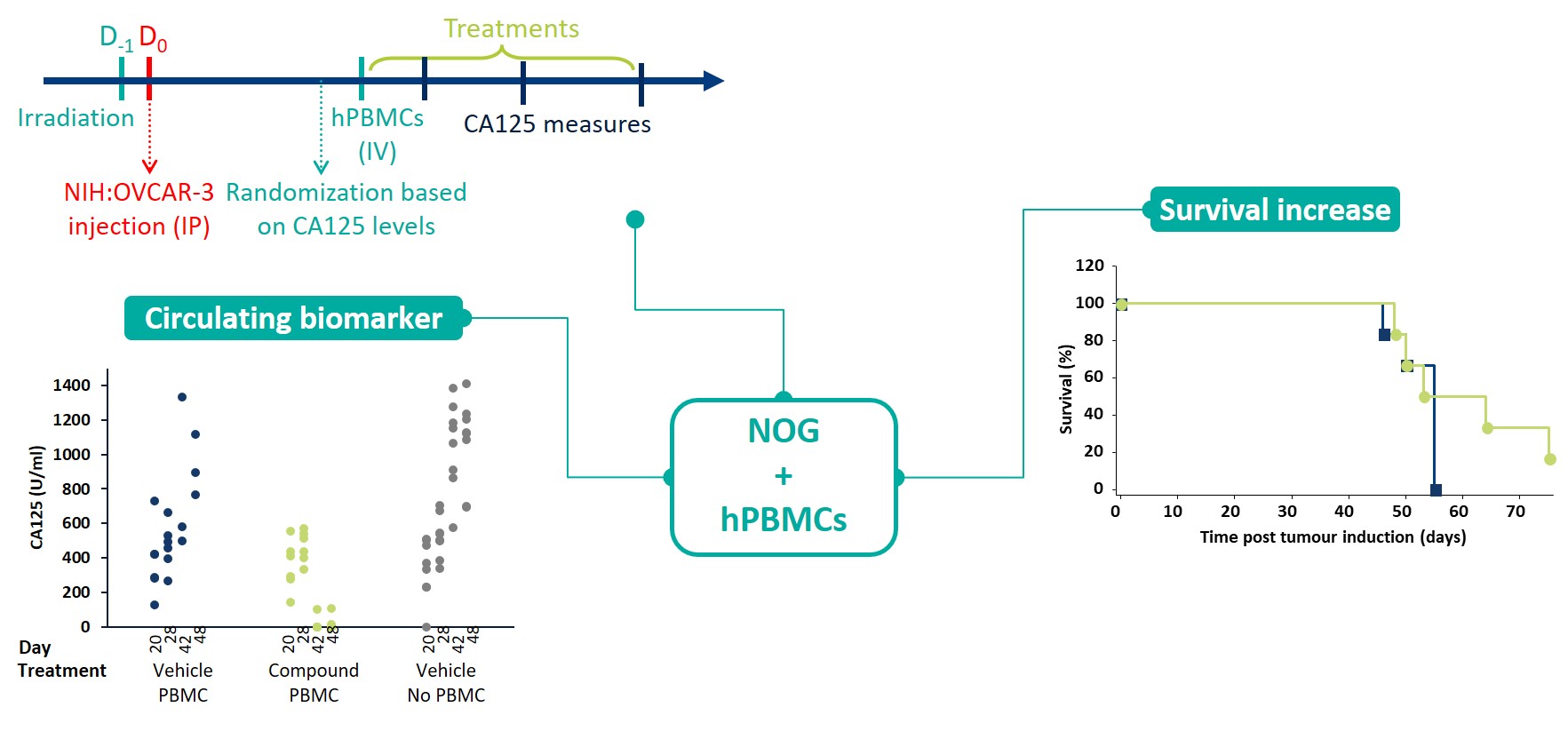
The NIH-OVCAR3 model was injected IP into humanized NOG-mice. The mice were treated with an anti-PD-1 immune checkpoint inhibitor bispecific, with tumor volume estimated from the level of circulating biomarker CA125. The test article successfully suppressed the levels of CA125 and improved survival a little.
FFPE Slide Samples
Slide samples are available from FFPE tumor blocks, for the following humanized tumor models. Details of the humanization, including mouse background, are available upon request.
Slides are free of charge, but you pay for shipping and handling. Alternatively, ship us your antibody and we will do the IHC on our end, to confirm expression of your target.
| Models | |
|---|---|
| A375 | Malignant melanoma |
| BT-474 Her2+ | Breast ductal carcinoma |
| DU145 | Prostate |
| HCC-1954 | Pancreas adenocarcinoma |
| HT-1376 | Urinary bladder carcinoma |
| HT-29 | Colorectal adenocarcinoma |
| K-562 | Chronic myelogenous leukemia |
| MOLM13 | Acute myeloid leukemia |
| MX-1 | Breast adenocarcinoma |
| NCI-H929 | Plasma cell myeloma |
| NIH :OVCAR-3 | Ovarian adenocarcinoma |
More information about our humanized models ?