SMPT : an in vivo design allowing screening of PDX tumors
Patient-derived xenograft (PDX) tumor models are now considered the gold standard for translational in vivo tumor modeling, yet they are slow to grow and not easily scaled up. There is considerable market demand for a solution that combines the translatability of PDX tumors and the cost-effectiveness of in vitro screening.
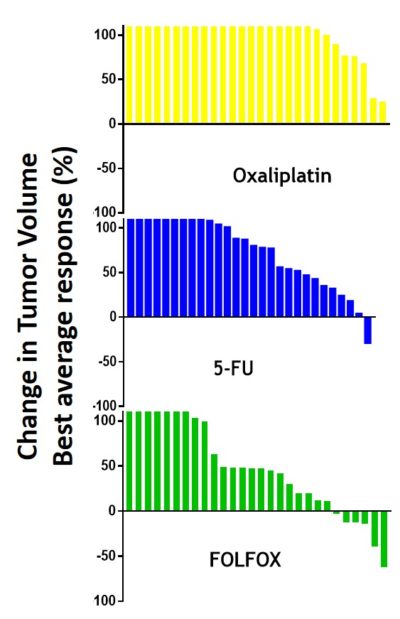
Oncodesign Services presents the Single Mouse Preclinical Trial (SMPT), where a range of selected PDX tumors are grafted into groups, but where each group consists of just one or two mice. This is similar to the mathematics of human clinical trials, where one patient represents one tumor. A waterfall plot of responders is generated as the read-out.
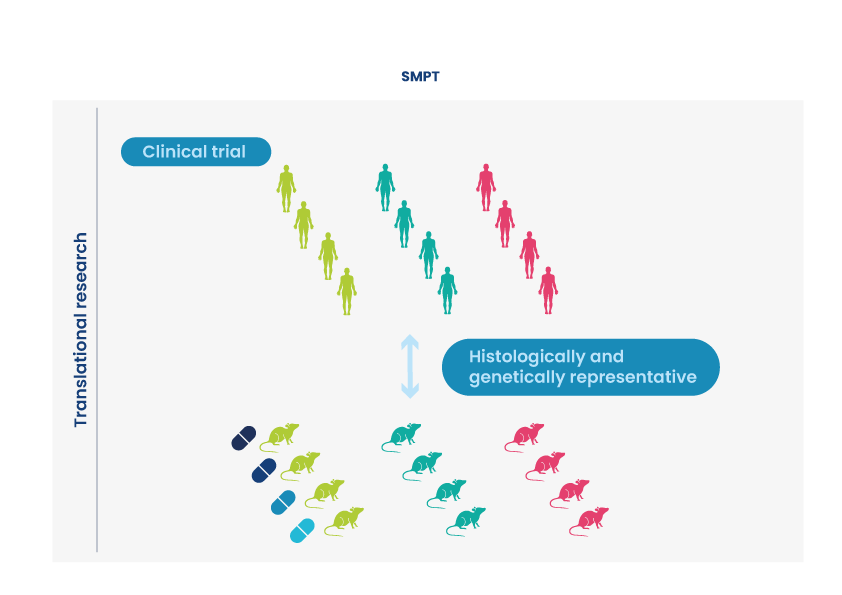
The SMPT is designed for drug developers who need to characterize:
- the range of responders across many indications
- the signature of response of drugs
- how responders change with drug combinations
Example:
27 colon PDX were injected into mice, with 3 of those subsequently enrolled in the trial, after confirmation of tumor growth. For the trial, one mouse with one distinct colon tumor was enrolled in each of 3 groups. The 3 groups were treated either with 5-FU and Oxaliplatin alone, or in combination. The resulting waterfall plot clearly shows the synergistic effect of the drug combination for most patients. This method can be used to screen PDX for drug combination strategies.